We have a new manuscript out in Progress in Retinal Eye Research, Inner-retinal changes in AMD: Evidence, mechanisms, and future perspectives.
Authors: Matt Trinh , Michael Kalloniatis , Bryan William Jones @bwjones.bsky.social, Glenn C Yiu, Enrico Borrelli, Lisa Nivison-Smith.
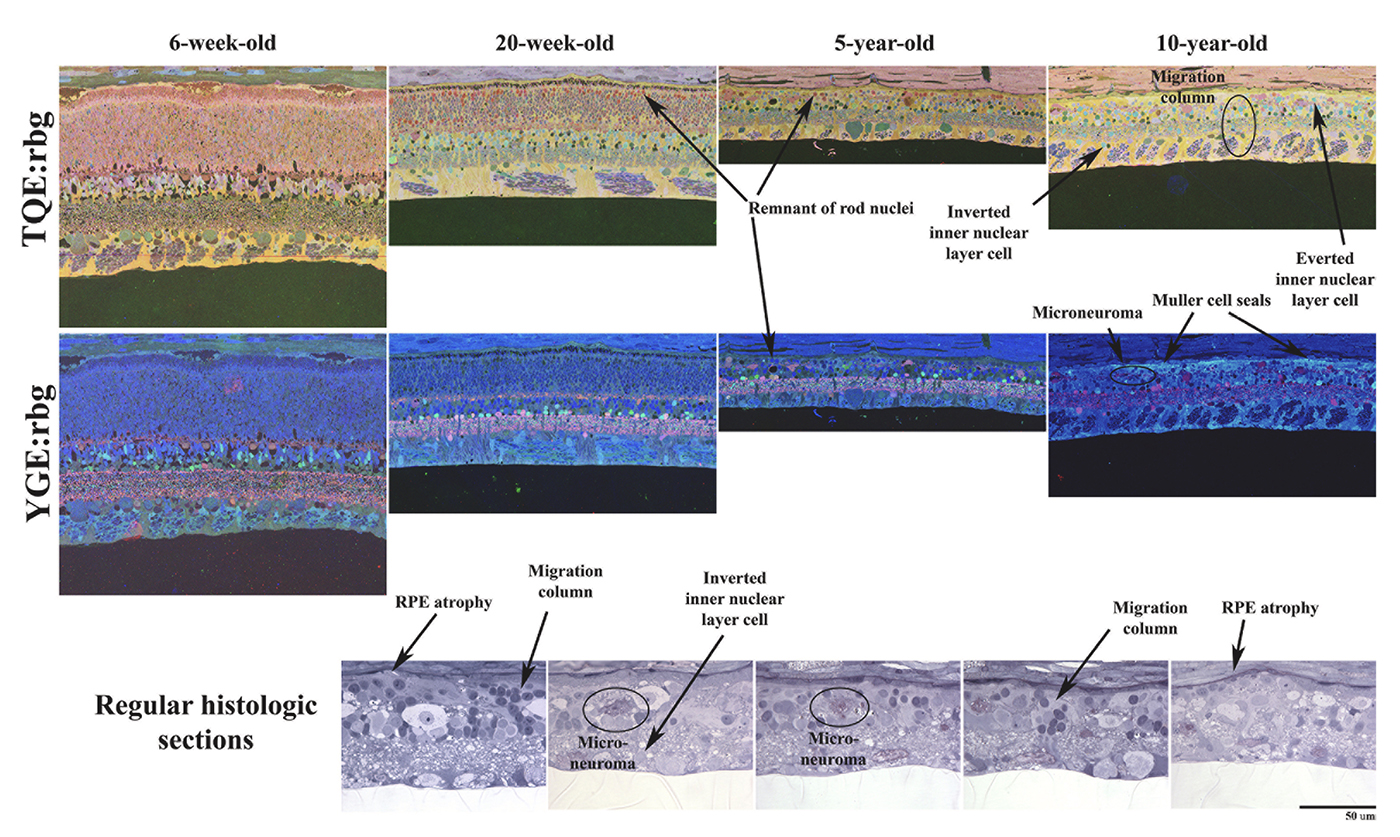
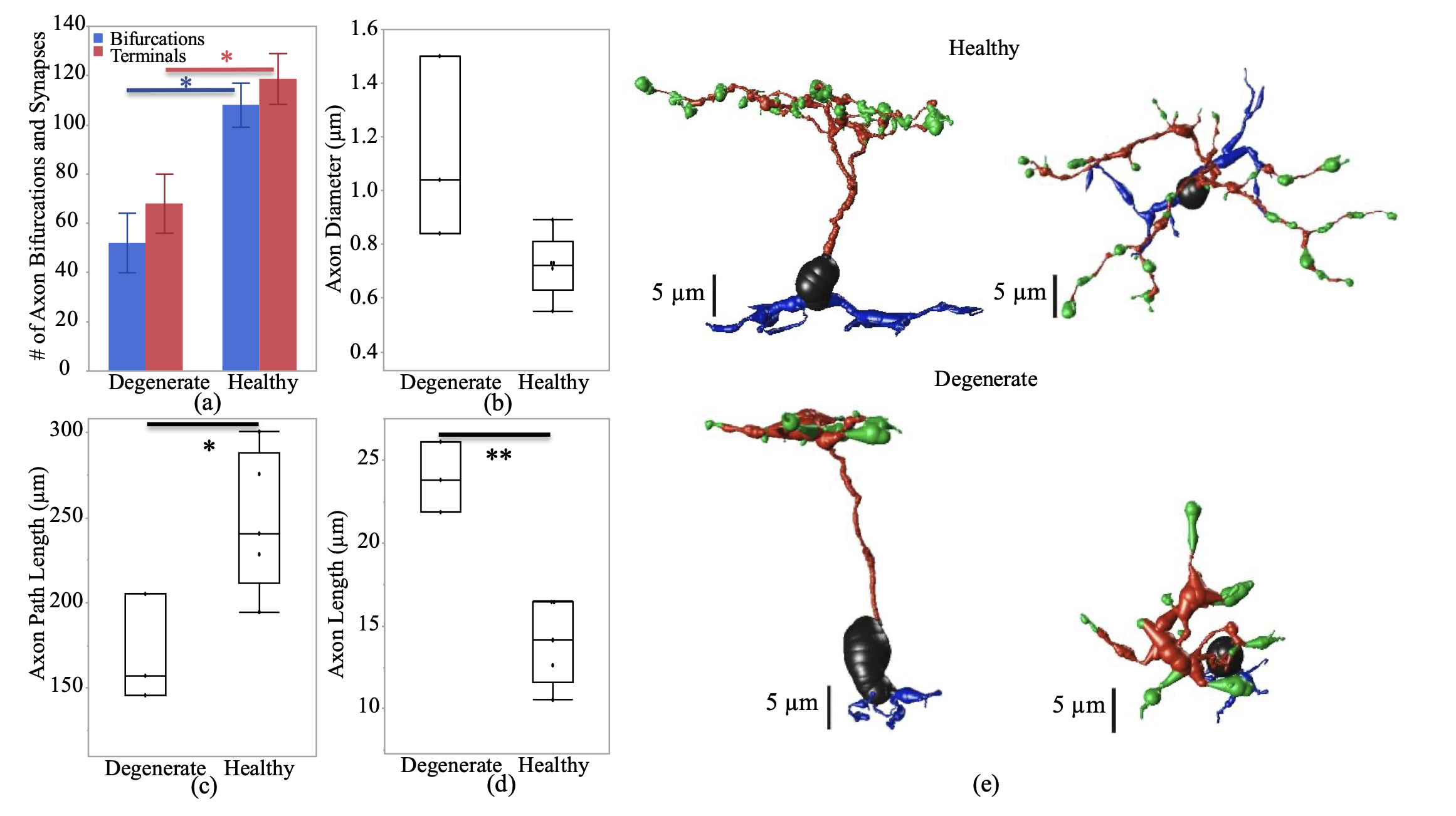
Abstract: Age-related macular degeneration (AMD) has traditionally been regarded as a disorder of the outer-retina and choroid, characterised by drusen accumulation, retinal pigment epithelium (RPE) dysfunction, and photoreceptor degeneration. However, increasing evidence of inner-retinal involvement across the AMD spectrum, with structural and functional compromise evident from the early stages of disease, challenges this paradigm. Advances in spatially optimised optical coherence tomography (OCT), OCT angiography (OCTA), and high-resolution histology have revealed neuronal, vascular, and glial alterations within the inner-retina that reshape our understanding of AMD pathogenesis. This review synthesises clinical and experimental evidence on inner-retinal changes in AMD, including layer-specific thinning, microvascular rarefaction, impaired neurovascular coupling, and reactive gliosis. Such changes frequently emerge in early AMD, may precede, parallel, or exacerbate outer-retinal degeneration, and are associated with visual dysfunction not fully explained by photoreceptor loss alone. Importantly, mechanistic interactions between inner- and outer-retinal pathology support a bidirectional model of neurodegeneration, wherein region-specific vulnerability is shaped by perfusion dynamics, metabolic demands, and structural connectivity throughout the retina. Recognition of these processes expands the potential for earlier diagnosis, refined monitoring, and novel therapeutic targeting. By integrating structural, functional, and systemic insights, this review reframes AMD as a multi-layer neurovascular disease and underscores the central role of inner-retinal integrity in future AMD research and management.